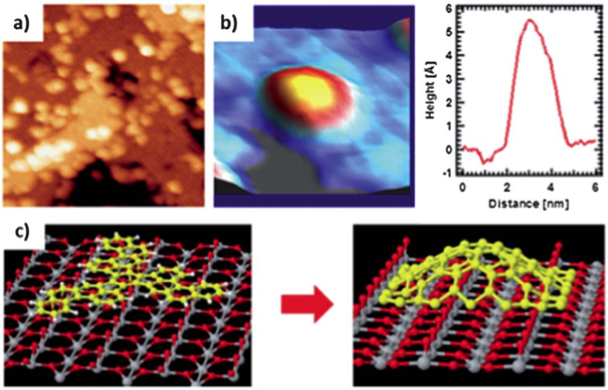
We evidence temperature-assisted (cyclo)dehydrogenation reactions of large polycyclic aromatic hydrocarbons (PAHs) for the paradigmatic case of C60H30, and the subsequent bottom-up formation of assembled nanostructures, such as nanodomes, on a dielectric substrate like the TiO2(110) surface. To this aim we have deposited, under ultra-high vacuum environment, submonolayer coverage of C60H30, and we have studied, by a combination of experimental techniques (STM, XPS and NEXAFS) and theoretical methods, the different chemical on-surface interaction stages induced by the increasing temperature. We show that, at room temperature, adsorbed molecules exhibit a weak interaction and freely diffuse on the surface. Nevertheless, a slight annealing induces a transition from this starting (meta)stable configuration towards strongly chemisorbed molecules, in which the perylene aromatics interact with the surface deforming the molecular structure and, as a consequence, surface diffusion is quenched. Higher annealing temperatures lead to partial dehydrogenation, in which the molecule loses some of the peripheral hydrogen atoms and the LUMO level spreads out, manifesting a net total energy gain. Further annealing, up to around 750K, ends ups into complete dehydrogenation. At these temperatures the fully dehydrogenated molecules link between them in a bottom-up coupling, forming nanodomes or fullerene-like monodisperse species. This work opens the door to the use of on-surface chemistry to generate new bottom-up tailored structures directly on high-K dielectric surfaces.
Full text in this link: Chemistry and Temperature-assisted Dehydrogenation of C60H30 Molecules on Ti02(110) surfaces, C. Sánchez-Sánchez, J. I. Martínez, V. Lanzilotto, G. Biddau, B. Gómez-Lor, R. Pérez, L. Floreano, M. F. López y J. A. Martín-Gago; Nanoscale, 5 (2013) 11058.