Recently, we have published a new work in Nature Communications:
Graphene functionalization with organics arises as a very promising route towards the development of graphene-based materials with tailored electronic properties. However, the high chemical inertness of graphene turns into very difficult task a controlled and selective covalent functionalization with aromatics, and most of the works performed up to the date report either electrostatic molecular adsorption or unruly functionalization by wet-chemical methodologies. A ruled covalent functionalization will provide added value to pristine graphene in order to develop applications in domains as important as band-gap engineering, creation of magnetism in graphene for applications in spintronics, fabrication of dielectrics, formation of nano-bio hybrid composites for improved biosensors or tailoring optical-plasmonic properties. In particular, following this strategy, biomolecules can be linked to the graphene surface, opening the door to biosensing, enzymatic biosensing, DNA sensing, bio-field-effect transistors, or immunosensing. In this way, we would like to emphasise their usage within state-of-the-art graphene field effect transistors (GFET). On the other hand, this protocol can be used as an easy way of introducing elements or magnetic nanotructures within the graphene surface, and is also compatible with other hetero-applications, in which one could combine the interesting graphene properties requiring low-sheet resistance, as solar cells, light-emitting diodes (LED), liquid-crystal displays (LCD), touch screens, with other targeted functionalities provided by the new groups.
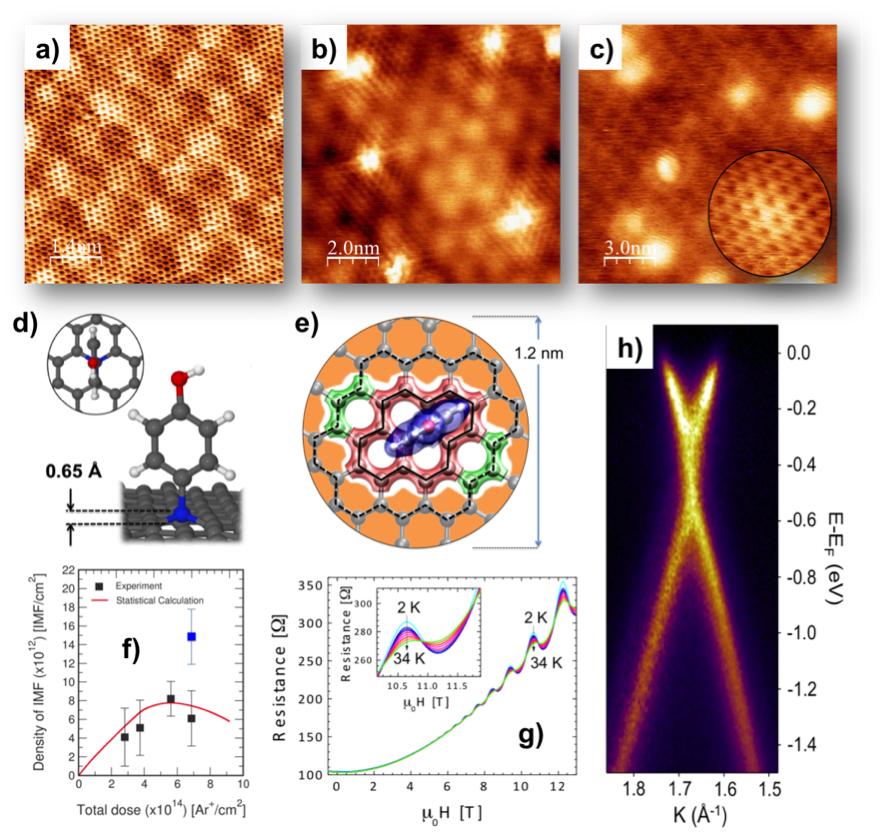
On this basis, we propose a new mechanism for promoting highly specific covalent bonding of any amino-terminated organic molecule and a description of the operating processes at the nanoscale. Excess of charge at carbon dangling bonds created on the graphene surface by low-energy ion-sputtering presents enhanced reactivity towards dehydrogenation reactions of molecules containing amino groups. The p-aminophenol molecules links covalently to the graphene via a selective oxidation of the amino group and subsequent integration of the nitrogen within the graphene network in a sp3-like configuration. Remarkably, the functionalized surfaces retain the electronic properties of pristine graphene, as unaltered Shubnikov-de-Haas oscillations and unmodified Dirac cone. This study opens the door for development of new graphene-based interfaces with effects in many different fields, among then nano-bio-hybrid composites, fabrication of dielectrics, plasmonic or spintronics as mentioned.